CATEGORY
Connected Devices and Wearables
Digital Technology that could disrupt the equipment market.
Beroe LiVE.Ai™
AI-powered self-service platform for all your sourcing decision needs across 1,200+ categories like Connected Devices and Wearables.
Market Data, Sourcing & Supplier Intelligence, and Price & Cost Benchmarking.
Schedule a DemoThe World’s first Digital Market Analyst
Abi, the AI-powered digital assistant brings together data, insights, and intelligence for faster answers to sourcing questions
Abi is now supercharged with GPT4 AI engine. Enjoy the ease of ChatGPT, now on Abi
Connected Devices and Wearables Suppliers
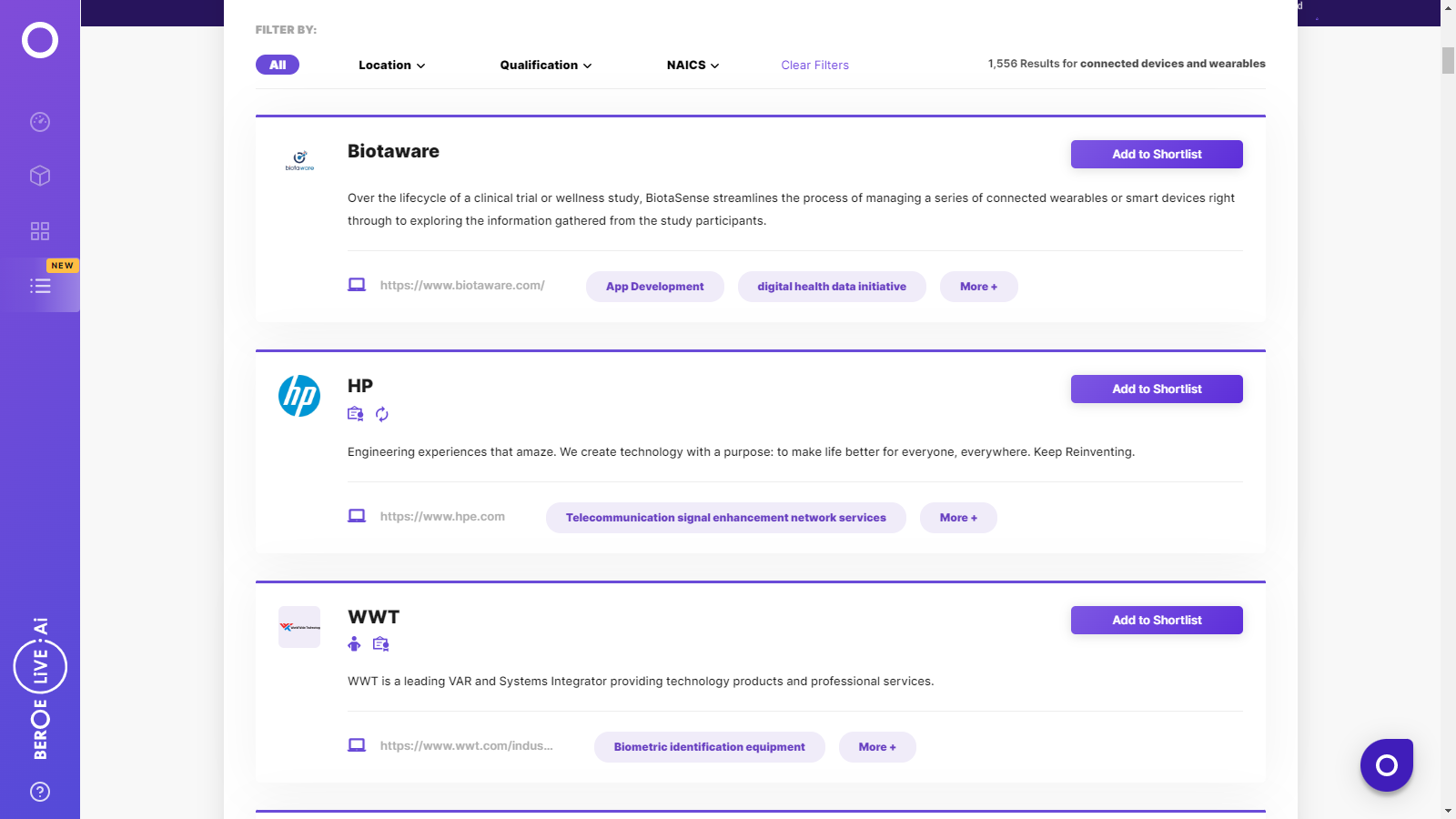
Find the right-fit connected devices and wearables supplier for your specific business needs and filter by location, industry, category, revenue, certifications, and more on Beroe LiVE.Ai™.
Schedule a Demo


Use the Connected Devices and Wearables market, supplier and price information for category strategy creation and Quaterly Business Reviews (QRBs)
Schedule a DemoConnected Devices and Wearables market report transcript
Global Market Outlook on Connected Devices and Wearables
The rapid growth in digital technology and the rise of connected health offer ways to develop innovative and differentiated patient services, speed up clinical trials, and analyze new data from connected devices to drive improved consumer insight and personalization and improve outcome.
Patient Outcome in Clinical Trials
- Improve efficiencies and reduce cost
- Advance prevention over treatment
- Promote individual ownership and control over personal health data, including the ability to share it with one's healthcare provider, caregivers or social network
- Improve communication and data exchange between patients and providers
- Create new incentives for a movement toward personal responsibility and engagement in one's own health
Success in personal connected health will be measured in a number of ways, including adoption of personal connected health tools and services.
Digital technology is an essential part of emerging solutions, engaging community health care resources—physicians, diagnostic facilities, labs, visiting nurse involved in Clinical Trials.It also provides additional benefits of expanding the geographic footprint outside traditional academic medical centers and democratizing trials by including patients and physicians who today are not part of the research process.
The Connected Health market will grow to a substantial size of more than $28 billion in 2017. In comparison with the global healthcare market, which is estimated to have a gigantic size of $6 trillion, Connected health devices represents 0.5%.
Connected Health Market Size (2016–2020)
- Connected Health is a new model for health management that puts the correct information in the correct hands at the correct time, so it is about engaging patients with their health through technology
- The personal connected health market is growing between 35 and 38% annual growth rate through 2022
- The NIH Strategic Plan for 2016–2020 incorporates the study of mHealth technologies and their ability to help prevent and treat illness as a research priority, with an objective of improving health outcomes, health care services, or health research
Technologies in Connected Health
- The technologies with the perceived most disruptive potential in the next five years is artificial intelligence and remote monitoring. Wearables, Internet of things (IoT) Virtual reality, 3D printing, and Block chain are next in the ranking, leaving 5G as the least disruptive
- Most mHealth publishers are rather new to the market, these established and successful stakeholders can serve as best-in-class or role models and pave the way for the new breed of digital intruders
- The popularity of health insurers as a distribution channel has been slowly increasing over the years, and has even overtaken “app stores” as the best distribution channel
Regulatory Landscape
Within the digital health ecosystem, software used in the healthcare setting, it will be important to consider whether any technology is a medical device under the relevant legislation and guidanceIn terms of data security and privacy, HIPAA, Safe Harbor, and EU Data Protection are the guidelines to follow, in terms of compliance.
- At the moment, there are no established standards specific to digital health and so, auditing suppliers for data risk is a necessary process
- Technology vendors use the necessary regulations as a guide to assure that they are utilizing good processes for managing their electronic records and other documents
- Due to the nature of the industry, evolving regulation will help drive adoption of mHealth-based research models
- The vital importance of doctor-patient confidentiality means these concerns will always be high on the list for any NHS technology
Interesting Reads:
Discover the world of market intelligence and how it can elevate your business strategies.
Learn more about how market intelligence can enable informed decision-making, help identify growth opportunities, manage risks, and shape your business's strategic direction.
Get Ahead with AI-Enabled Market Insights Schedule a Demo Now
