The major crux of pharmaceutical production has shifted toward contract manufacturers and specialized suppliers. Companies increasingly rely on external partners for active pharmaceutical ingredient (API) synthesis, formulation development, spanning monoclonal antibodies (mAbs), antibody-drug conjugates (ADCs), recombinant proteins, and cell therapies, as well as commercial manufacturing. This transformation has reshaped procurement’s role. However, procurement strategies remain overly cost-focused, creating structural risk amid intensifying regulatory scrutiny, rising inspections, and high Form 483 rates. Today, procurement must also consider supplier quality, regulatory history, supply resilience, and long-term partnership potential. It therefore must shift to regulatory-risk–integrated models, prioritizing high-compliance partners and real-time regulatory intelligence. Products approved under strict regulatory frameworks must be manufactured in facilities with strong quality systems and robust documentation practices.
As a result, procurement teams are increasingly confronted with a critical strategic question: Should companies pay a premium to source from suppliers with stronger regulatory track records? Answering this question requires a deeper understanding of regulatory risk, supplier maturity, and the long-term financial consequences of supply disruptions.
Introduction
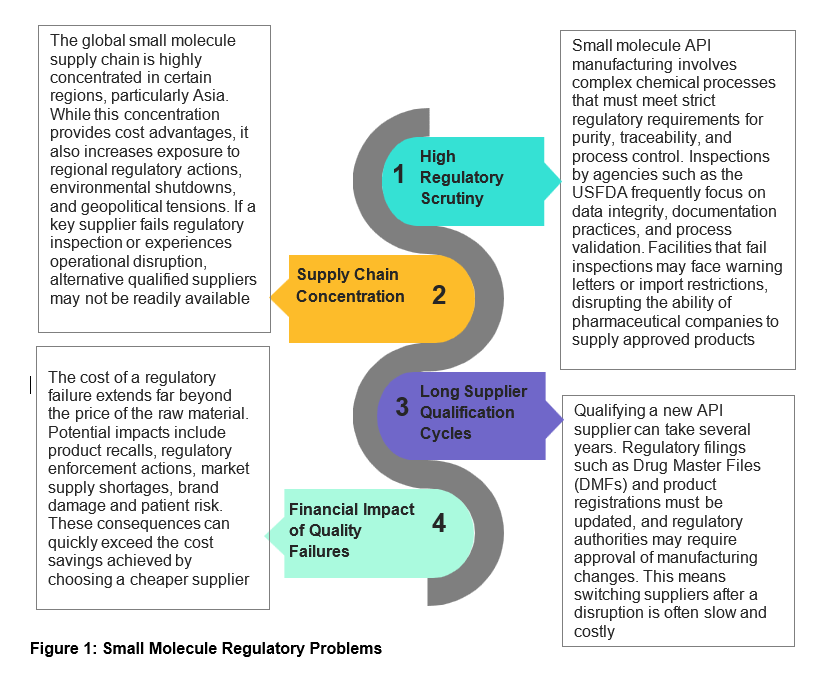
Small-molecule drugs continue to dominate the pharmaceutical landscape, accounting for a majority of global prescriptions and treatments. The manufacturing ecosystem for small molecules has evolved over the past few years. Regulatory agencies have tightened their expectations, and inspections have become more data-driven and global in scope. Observations such as warning letters, import alerts, or consent decrees can severely disrupt supply chains and market access. Similarly, large-molecule drugs account for ~40% of global pharmaceutical revenue. Biologics outsourcing to contract development and manufacturing organizations (CDMOs) has accelerated, and this shift makes supplier selection strategically critical. Variability in cell culture, purification, sterility, and cold-chain logistics introduces multiple risk points across the drug lifecycle.
Procurement, however, prioritizes cost savings and working capital efficiency over regulatory robustness. This bias toward low-cost suppliers can undervalue good manufacturing practice (GMP) maturity, quality systems, and inspection track records. Consequently, short-term savings are frequently offset by long-term risks, such as supply disruptions, regulatory actions, and recalls. Hence, paying a 5–15% premium for compliant suppliers is economically justified to ensure supply resilience, patient safety, and long-term cost efficiency.
Cost vs Regulatory Risk in Pharma Procurement
Pharmaceutical procurement teams frequently face tension between cost optimization and supply assurance. For example, when a manufacturing site fails to meet regulatory requirements, authorities may restrict product imports, leading to drug shortages and commercial losses. The implications are particularly significant for companies supplying regulated markets such as the US and Europe.
| Type of Molecule | Official Action Indicated (OAI) | Form 483 | European Medicines Agency (EMA) | Remediation | Revenue Risk |
| Small Molecule | 10-12% | 4.0–5.2 | 18-20% | $5-15M | $50-300M |
| Large Molecule | 14-16% | 5.0–6.5 | 24–26% | $20M–$75M | $200M–$1B+ |
Table 1: Data and Regulatory Enforcement Trend Outlook
Despite these risks, many procurement organizations still prioritize short-term cost reductions due to budget pressures and performance metrics focused on price savings. This creates a strategic dilemma: Should procurement absorb higher costs upfront to reduce regulatory risk? The tension between cost-driven procurement and regulatory risk has intensified as biologics scale and oversight tightens, driven by three core dynamics:
Regulatory complexity: Biologics rely on inherently variable cell-based systems where tight control of process parameters is critical to product quality. International Council for Harmonisation (ICH) Q8–Q10 frameworks demand advanced process understanding, risk management, and robust quality systems. Aseptic processing and sterility assurance add further vulnerability, with contamination events causing prolonged production disruptions. Advanced modalities like ADCs introduce additional challenges, including containment, linker chemistry control, and drug-to-antibody ratio (DAR) consistency.
Procurement pressure: CDMO and API costs account for 30–50% of biologics Cost of Goods Sold (COGS), driving aggressive cost-reduction mandates. Short-term contracts disincentivize supplier investment in long-term quality infrastructure, weakening regulatory maturity.
Operational risk: Non-compliant suppliers face escalating inspections, warning letters, and import restrictions, disrupting global supply. Remediation cycles can last 12–24 months, delaying approvals and halting commercial production. Ultimately, short-term cost savings are frequently outweighed by significant financial and operational risks, including supply disruptions and high-cost recalls. Table 2 summarizes the estimated cost impact of compliance failure in biologics manufacturing.
| Compliance Failure | Estimated Financial Impact |
| FDA Warning Letter (CDMO remediation) | $10M – $50M direct; $50M–$500M lost revenue |
| Import Alert (US. market exclusion) | $100M–$1B+ annual revenue at risk |
| Class I Biologics Recall | $100M–$600M total cost |
| Manufacturing Shutdown (12–24 months) | $50M–$500M per program |
| Biologics Launch Delay (per year) | $300M–$500M lost NPV |
Table 2: Estimated cost impact of compliance failure in biologics manufacturing
What Criteria Define High-Quality and Compliant Pharmaceutical Suppliers?
To determine whether paying a premium for regulatory strength is justified, procurement teams must evaluate supplier options against a set of structured criteria for small and large molecules.
| Regulatory Compliance Track Record | Suppliers should be assessed based on their inspection history with major regulatory agencies such as the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA). Metrics may include: Number of inspections conducted, Warning letters or enforcement actions, Import alerts or product bans, Time required to close regulatory observations | A consistent record of successful inspections indicates mature quality systems and regulatory discipline. |
| Quality System Maturity | Suppliers should be assessed based on their inspection history with major regulatory agencies such as the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA). Metrics may include: Number of inspections conducted, Warning letters or enforcement actions, Import alerts or product bans, Time required to close regulatory observations | A consistent record of successful inspections indicates mature quality systems and regulatory discipline. |
| Operational Reliability | Procurement should evaluate a supplier’s ability to maintain consistent production and delivery performance, including: Capacity utilization levels, Redundancy in manufacturing lines, Raw material sourcing stability, Track record of on-time delivery | Operational reliability reduces the risk of supply shortages. |
| Market Access Alignment | Suppliers must be capable of supporting regulatory requirements in target markets. For example, suppliers serving regulated markets such as the US and Europe must maintain compliance with stringent regulatory frameworks. | Suppliers that regularly serve these markets are better equipped to support global pharmaceutical companies. |
| Total Cost of Ownership (TCO) | Regulatory strength often correlates with the maturity of a supplier’s quality management system, including: Data integrity practices, Change control procedures, Deviation management, Process validation, and documentation standards | A comprehensive cost model can reveal that lower-priced suppliers may carry higher long-term costs. |
Table 3: Criteria, Assessment, and Benefits of Regulatory Scrutiny
Procurement organizations require a structured, evidence-based framework to evaluate biologics suppliers that extends beyond price and capacity. Table 2 represents the essential dimensions of regulatory maturity assessment.
| Description & Indicators | |
| Regulatory Inspection History | FDA, EMA, and Pharmaceuticals and Medical Devices Agency (PMDA) inspection frequency, Form 483 observation trends, warning letter history, and import alert status |
| GMP Maturity Score | ICH Q10 pharmaceutical quality system implementation, Corrective and Preventive Action (CAPA) effectiveness, change control rigor, deviation closure rates |
| Quality Management Systems (QMS) | Electronic QMS platform, document control, supplier qualification procedures, batch record integrity |
| Manufacturing Redundancy | Multi-site capability, technology transfer readiness, contingency production capacity for critical products |
| Digital Traceability | Electronic batch records, Manufacturing Execution System (MES) integration, real-time process analytics, data integrity compliance (21 CFR Part 11) |
| Long-Term Cost Stability | Historical pricing stability, investment trajectory in facilities and equipment, and financial health indicators |
Table 4: Criteria, Description, and Indicators for Biologics Regulatory Assessment
Procurement Strategies in Pharma Sourcing
Procurement organizations have several strategic options when addressing regulatory risk in supplier selection for small and large molecules.
Balanced Cost and Compliance Model: This model balances cost considerations with regulatory strength. Procurement assigns weighted scores to suppliers based on price, regulatory performance, quality systems, and supply reliability. Suppliers with strong regulatory records may receive higher scores even if their prices are moderately higher.
Regulatory-Preferred Supplier Strategy: Procurement deliberately prioritizes suppliers with strong regulatory track records and high-quality standards. These suppliers may command price premiums but offer greater reliability and reduced risk of supply disruptions.
Dual-Sourcing Strategy: Companies maintain relationships with both premium regulatory suppliers and lower-cost secondary suppliers. This approach allows procurement to balance cost efficiency with supply security. However, it requires careful regulatory planning and supplier qualification.
These partnerships reduce disruption risk, accelerate approvals, and avoid costly remediation cycles. Ultimately, lifecycle cost savings from regulatory-first models consistently outweigh upfront price premiums.
Why Should Organizations Prioritize Regulatory Credentials In Supplier Selection?
Several industry trends support the case for prioritizing regulatory strength in supplier selection.
Increasing Regulatory Enforcement: Regulatory agencies are conducting more targeted inspections and leveraging advanced analytics to identify compliance risks. Facilities with weak quality systems are more likely to face enforcement actions. Companies sourcing from such suppliers may experience unexpected disruptions if authorities restrict imports or suspend manufacturing operations. ICH Q9 risk frameworks show that low-compliance suppliers carry the highest probability and severity of failure. However, procurement rarely quantifies supplier risk using these principles, creating a governance gap. Market analysis estimates compliance-related disruptions add 12–18% to lifecycle COGS.
Rising Cost of Supply Disruptions: Drug shortages can have severe financial and reputational consequences. When supply interruptions occur, companies often must source materials from alternative suppliers at significantly higher costs. In extreme cases, companies may temporarily lose market share or face contractual penalties for failing to meet supply commitments.
Long-Term Supplier Partnerships: Suppliers with strong regulatory capabilities often invest heavily in quality infrastructure, advanced analytics, and workforce training. These investments support long-term collaboration and innovation. Pharmaceutical companies working with such suppliers benefit from higher transparency, stronger process control, and improved regulatory readiness.
Regulatory Complexity in Global Markets: Pharmaceutical companies increasingly operate across multiple regulatory jurisdictions. Suppliers capable of supporting global regulatory submissions provide strategic advantages. These suppliers often maintain well-documented processes and regulatory expertise that simplify product registration and lifecycle management.
Pharma Regulatory Case Studies
Regulatory alignment has emerged as a primary determinant of manufacturing resilience, shaping both approval timelines and long-term supply continuity. As regulatory agencies converge on stricter, harmonized expectations, manufacturers are required to embed quality-by-design, data integrity, and lifecycle control into core operations. This shift moves compliance from a reactive obligation to a proactive capability that differentiates suppliers in both small and large molecule markets. Listed in Table 5 are case studies that illustrate how varying levels of regulatory maturity translate directly into commercial outcomes and supply reliability, indicating that regulatory maturity is not a cost premium but is a measurable driver of long-term economic value.
| Regulatory Failure & Recovery – Small Molecule | Import alert Impact on Indian API Manufacturer due to data integrity lapses, and inadequate out-of-specification (OOS) investigations. The manufacturer faced 12–18 months of disruption across multiple Abbreviated New Drug Application (ANDA)-linked products, loss of US market access, shifting demand to alternate suppliers, and an estimated $100M–$250M revenue impact |
| Regulatory Leadership – Small Molecule | Pfizer’s continuous investment in digital batch records, real-time release testing (RTRT), and integrated quality systems has enabled low repeat 483 rates, faster regulatory approvals for post-approval changes |
| Regulatory Failure – Large Molecule | WuXi Biologics faced heightened US regulatory scrutiny, due to geopolitical risk affecting customer confidence, leading to sponsors initiating re-shoring strategies, delays in tech transfer and regulatory filings, and an estimated multi-$100M pipeline risk exposure |
| Regulatory Leadership – Large Molecule | Samsung Biologics performance and continued zero warning letter track record, expansion of multi-plant Songdo mega-site, and strong adherence to Annex 1 sterile requirements lead to being a preferred partner for top 20 pharma biologics outsourcing, and accelerated approvals due to inspection confidence |
Table 5: Small and Large Molecules Supplier Case Studies
Recommendations and Next Steps
Strategies to Overcome Regulatory Problems
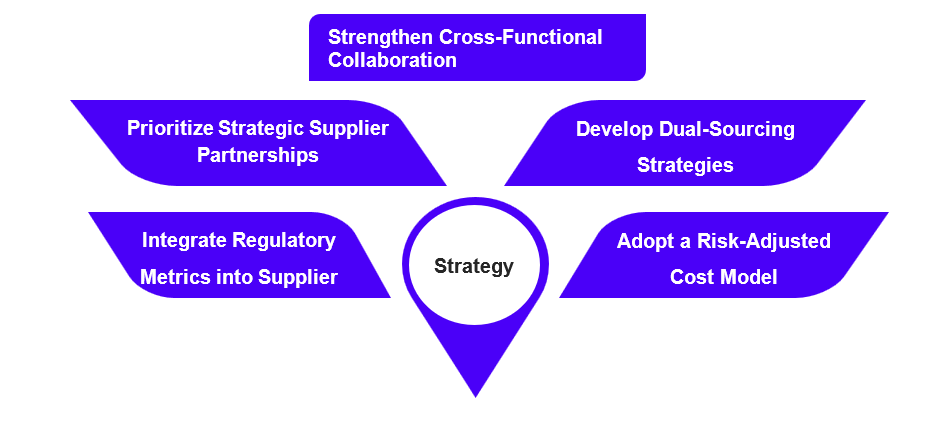
Procurement teams should incorporate regulatory performance indicators into supplier evaluation frameworks. They should assign a 30–40% weighting to regulatory risk in supplier evaluation models and develop real-time monitoring of global inspection outcomes, 483 trends, warning letters, and import alerts. They should track CDMO investments and geopolitical risks impacting supply chains and leverage platforms such as Veeva Systems and TraceLink to enable proactive risk detection.
Organizations should move beyond simple price comparisons and adopt total cost of ownership models that account for regulatory risk and supply disruption costs. Cross-functional decision-making ensures that sourcing choices align with corporate quality and compliance objectives.
Long-term partnerships with regulatory-strong suppliers can improve supply reliability and support product lifecycle management. Suppliers should include quality investment commitments, inspection-performance KPIs, and multi-site contingency provisions, while aligning incentives through joint quality improvement programs and technology transfer rights. These partnerships may involve collaborative planning, capacity commitments, and shared quality initiatives.
Procurement should collaborate closely with quality, regulatory, and supply chain teams to evaluate supplier risk and develop comprehensive sourcing strategies. Where feasible, companies should qualify multiple suppliers for critical materials. Maintaining both primary and secondary suppliers reduces reliance on a single manufacturing site.
Call to Action and Implementation Timeline
The pharma supply chain is a complex, highly regulated ecosystem in which the quality decisions embedded at the point of supplier selection propagate forward, through manufacturing, regulatory approval, commercial launch, and long-term lifecycle management. Procurement leaders should recognize this reality and restructure sourcing strategies and accordingly build supply chains that are not merely cheaper to operate in the present but be resilient against the regulatory and operational shocks that will define competitive advantage in the future.
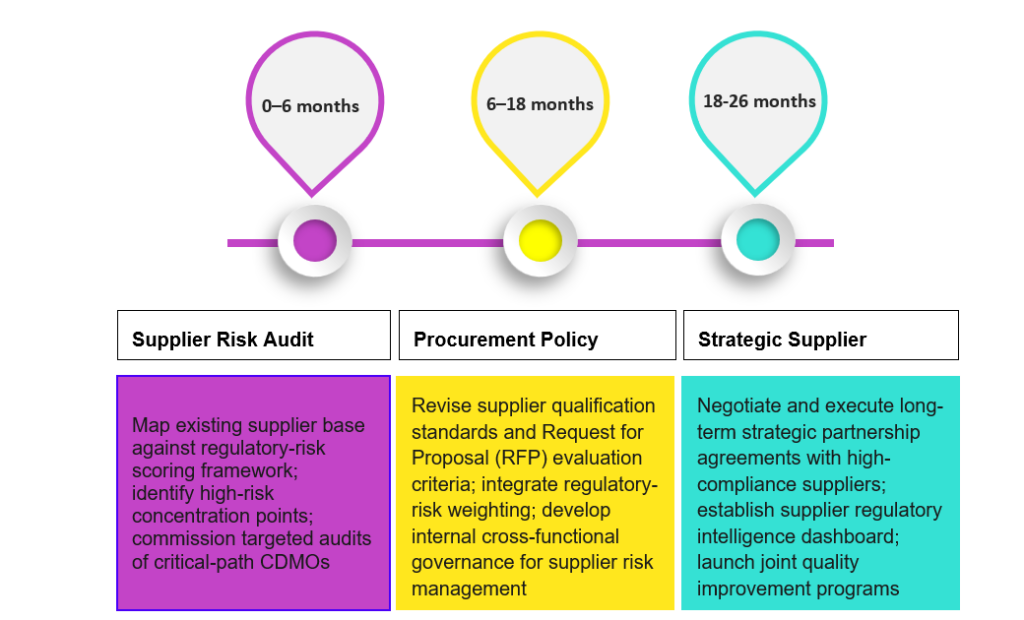
Figure 4: Actionable and Implementation Timelines
What Does Regulatory-First Procurement Deliver in Long-Term Value?
The pharmaceutical industry is entering a period where supply chain resilience and regulatory compliance are becoming as important as cost efficiency. For small molecule manufacturing, where complex chemical processes and stringent regulatory expectations intersect, supplier selection decisions carry significant strategic implications. Similarly, in large molecule manufacturing, the complexity of cell-based manufacturing, the sensitivity of biological products to process variation, and the expanding reach of global regulatory enforcement have collectively transformed regulatory maturity from a compliance obligation into a strategic commercial asset. Procurement organizations must therefore rethink traditional cost-focused sourcing models and should adopt risk-adjusted frameworks that incorporate regulatory performance as a core decision criterion.
The premium paid by companies for regulatory strength represents an investment in supply security, product quality, and patient safety. By aligning procurement strategies with regulatory and operational priorities, pharmaceutical companies can build more resilient supply chains and sustain long-term competitiveness in an increasingly complex global market. While paying a 5–15% premium for regulatory strong suppliers may appear costly in the short term, the long-term benefits often outweigh the expense. When total lifecycle cost is the unit of analysis, regulatory-first procurement is not a cost center, but rather a value-creation strategy.
References
U.S. Food and Drug Administration, “Inspections, Compliance, Enforcement, and Criminal Investigations”
World Health Organization, “WHO Guidelines on Good Manufacturing Practices for Pharmaceutical Products”
United States Pharmacopeia, “Global API Supply Chain Intelligence Report,” 2023.
IQVIA, “Global Medicine Spending and Usage Trends,” 2023.
U.S. Food and Drug Administration, “Warning Letters Database”
IQVIA Institute for Human Data Science, “Global Medicine Spending and Usage Trends: Outlook to 2028,” IQVIA Institute Report, Apr. 2024.
U.S. Food and Drug Administration, “Drug Establishment Inspection Data,” FDA Inspection Database, 2023.
Deloitte Centre for Health Solutions, “Pharmaceutical Supply Chain Risk: Quantifying the Cost of Manufacturing Disruptions,” Deloitte Insights, 2023.
Evaluate Pharma, “World Preview 2024, Outlook to 2030,” Evaluate Ltd., 2024.
BioPharma Reporter, “Monoclonal Antibody Market: Global Revenue and Pipeline Analysis 2024,” William Reed Business Media, 2024.
Grand View Research, “Biologics Contract Development and Manufacturing Organization (CDMO) Market Size, Share & Trends Analysis Report,” Grand View Research Inc., 2024.
U.S. Food and Drug Administration, “21 CFR Part 600: Biological Products: General,” Code of Federal Regulations, 2024.
International Council for Harmonisation, “ICH Q8(R2): Pharmaceutical Development; Q9(R1): Quality Risk Management; Q10: Pharmaceutical Quality System,” ICH Guidelines, 2023.
U.S. Food and Drug Administration, “Guidance for Industry: Manufacturing Considerations for Pivotal Studies and Commercial Manufacturing of Antibody-Drug Conjugates,” FDA Center for Drug Evaluation and Research, 2024.
McKinsey & Company, “Winning in Biologics: The CDMO Opportunity,” McKinsey Life Sciences Practice, 2023.
Pharmaceutical Technology, “The True Cost of a Warning Letter: Financial and Operational Consequences for Biologics CDMOs,” Pharma Tech, 2023.
Parenteral Drug Association (PDA), “PDA Survey Report: CDMO Quality and Compliance Risk in Biopharmaceutical Manufacturing,” PDA Technical Report, 2023.
U.S. Food and Drug Administration, “Enforcement Reports and Product Recalls: Biological Products 2018–2023,” FDA MedWatch, 2024.
Reuters, “WuXi Biologics and Geopolitical Supply Chain Risk: Lessons for Biologics Procurement,” Reuters Life Sciences Analysis, 2024.
Samsung Biologics, “Corporate Sustainability and Quality Report 2023,” Songdo, South Korea, 2023
Roche Group, “Roche Annual Report 2023: Manufacturing Quality and Supply Chain Resilience,” F. Hoffmann-La Roche AG, Basel, Switzerland, 2024.
IQVIA, “Total Cost of Ownership in Biologics Supply Chains: A Compliance-Weighted Analysis,” IQVIA White Paper, 2024.
Author
Related Reading

29 Apr, 2026
The Evolving Role of Contract Workers in Workforce Planning in 2026: Technology, Compliance, and Procurement Implications